Products & Services
Solutions for GPP Compliance
Temperature Monitoring, Temperature Mapping and Calibration Services.
5nines Technologies introduces PharmaScout, the first in its Scout range of wireless solutions, designed to remove the entire legal burden of GPP Rules relating to temperature monitoring, temperature mapping and calibration of equipment used for monitoring temperature sensitive pharmaceutical products.

Good Pharmacy Practice dictates that temperature devices and sensors are calibrated annually. Devices that gradually lose accuracy over time can lead to a false sense of security, place temperature sensitive pharmaceutical products and patient safety at risk and increase costs due to unnecessary stock write-off.
GPP Rules require an annual temperature mapping exercise for any space allocated for the storage and handling of pharmaceutical products with a specified labelled storage temperature. A mapping study establishes the temperature distribution within the zone being mapped and locates hot and cold spots.
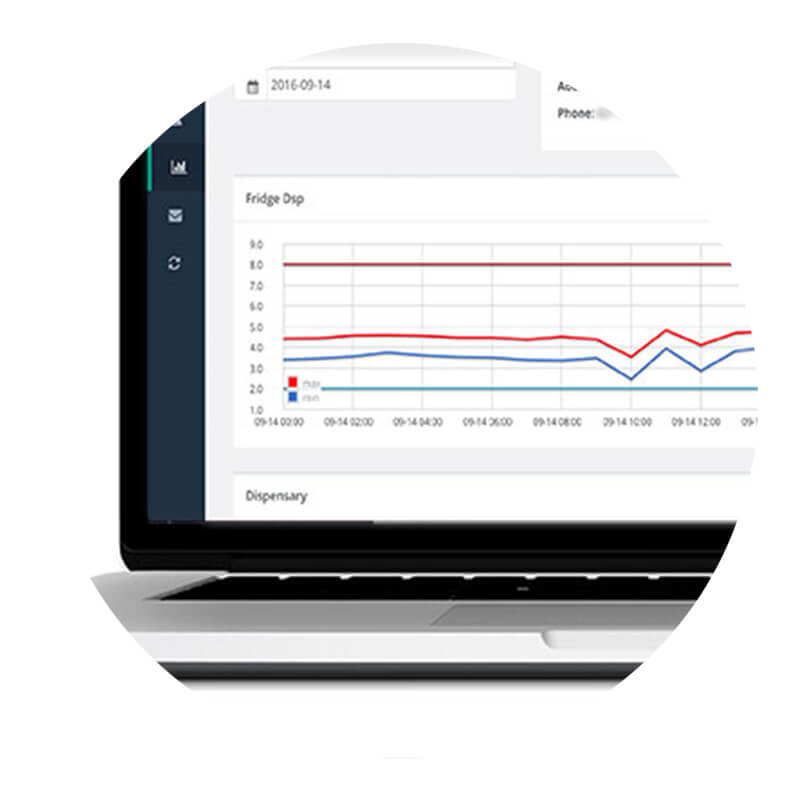
Automated temperature monitoring systems are easy to use, allowing the user the convenience to monitor and reliably document temperature information. These systems offer an accurate and cost effective solution to overcoming the GPP legal obligations for reporting, alarming, traceability, and good medication quality and safety management practices.